
The Miracle of Life: Carbohydrates, Proteins, Lipids & Nucleic Acids video can be viewed at (3:28). In a dehydration reaction, water is removed as two monomers are joined together. CarbonName: Carbon: Atomic Number: 6: Atomic Symbol: C : Atomic Weight: 12.0107 : Phase. Helps cells keep their shape, makes up muscles, speeds up chemical reactions, carries messages and materialsĬarbon, hydrogen, oxygen, nitrogen, phosphorusĬontains instructions for proteins, passes instructions from parents to offspring, helps make proteinsĬarbohydrates, proteins, and nucleic acids are large molecules (macromolecules) built from smaller molecules (monomers) through dehydration reactions. FactsRead key information and facts about element. Stores energy, forms cell membranes, carries messagesĬarbon, hydrogen, oxygen, nitrogen, sulfur Provides energy to cells, stores energy, forms body structures carbon, Nonmetallic chemical element, chemical symbol C, atomic number 6. Each element has a symbol, which is one or two letters. The periodic table lists the elements in order of increasing atomic number. Each element is identified by the number of protons in its atoms. Humans need 1018 milligrams of iron each day. There are 118 elements on the periodic table. They were given the name halogen, from the Greek roots hal - (salt) and - gen (to produce), because they all. The halogen elements are fluorine (F), chlorine (Cl), bromine (Br), iodine (I), astatine (At), and tennessine (Ts). Haemoglobin carries oxygen from our lungs to the cells, where it is needed for tissue respiration. halogen, any of the six nonmetallic elements that constitute Group 17 (Group VIIa) of the periodic table. A lot of this is in haemoglobin, in the blood. The average human contains about 4 grams of iron. You can compare the four types in Table below. Iron is an essential element for all forms of life and is non-toxic. In fact, there are nearly 10 million carbon-based compounds in living things! However, the millions of organic compounds can be grouped into just four major types: carbohydrates, lipids, proteins, and nucleic acids. This property allows carbon to form a huge variety of very large and complex molecules. Without carbon, life as we know it could not exist. Carbon is the main element in organic compounds, so carbon is essential to life on Earth. The International Union of Pure and Applied Chemistry (IUPAC) confirmed the names of elements 113, 115, 117, and 118 as: Nihonium (symbol Nh), for element 113. Organic compounds make up the cells and other structures of organisms and carry out life processes. The periodic table is a masterpiece of organised chemical information and the evolution of chemistry's periodic table into the current form is an astonishing achievement.  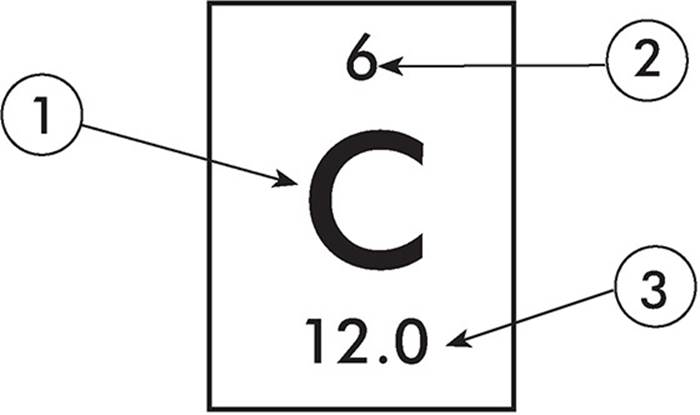
Why is carbon so basic to life? The reason is carbon’s ability to form stable bonds with many elements, including itself. A compound found mainly in living things is known as an organic compound.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
